Abstract
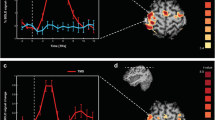
Being able to accurately quantify the hemodynamic response function (HRF) that links the blood oxygen level dependent functional magnetic resonance imaging (BOLD-fMRI) signal to the underlying neural activity is important both for elucidating neurovascular coupling mechanisms and improving the accuracy of fMRI-based functional connectivity analyses. In particular, HRF estimation using BOLD-fMRI is challenging particularly in the case of resting-state data, due to the absence of information about the underlying neuronal dynamics. To this end, using simultaneously recorded electroencephalography (EEG) and fMRI data is a promising approach, as EEG provides a more direct measure of neural activations. In the present work, we employ simultaneous EEG-fMRI to investigate the regional characteristics of the HRF using measurements acquired during resting conditions. We propose a novel methodological approach based on combining distributed EEG source space reconstruction, which improves the spatial resolution of HRF estimation and using block-structured linear and nonlinear models, which enables us to simultaneously obtain HRF estimates and the contribution of different EEG frequency bands. Our results suggest that the dynamics of the resting-state BOLD signal can be sufficiently described using linear models and that the contribution of each band is region specific. Specifically, it was found that sensory-motor cortices exhibit positive HRF shapes, whereas the lateral occipital cortex and areas in the parietal cortex, such as the inferior and superior parietal lobule exhibit negative HRF shapes. To validate the proposed method, we repeated the analysis using simultaneous EEG-fMRI measurements acquired during execution of a unimanual hand-grip task. Our results reveal significant associations between BOLD signal variations and electrophysiological power fluctuations in the ipsilateral primary motor cortex, particularly for the EEG beta band, in agreement with previous studies in the literature.







Similar content being viewed by others
Data Availability
The data collected for the purpose of this work are currently being curated and will be made available upon reasonable request (pending approval from our funding agencies and research institution).
Code Availability
The code that supports the findings of this study can be downloaded from the GitHub repository https://github.com/prokopis.
Notes
We considered the gamma density function given by \(\text{h}\left(\text{t};\uptau ,\upsigma \right)=\left\{\begin{array}{c}\text{exp}\left\{-\text{t}\sqrt{\upsigma \bullet\uptau } \right\}{\left(\frac{\text{e}\bullet\uptau }{\uptau }\right)}^{\sqrt{\uptau /\upsigma }}, \tau >0\\ 0, \tau <0\end{array}\right.\)where and control the location of the peak and width (dispersion), respectively (Hossein-Zadeh et al. 2003).
References
Abreu R, Leal A, Figueiredo P (2018) EEG-informed fMRI: a review of data analysis methods. Front Hum Neurosci 12:1–23. https://doi.org/10.3389/fnhum.2018.00029
Allen PJ, Polizzi G, Krakow K, Fish DR, Lemieux L (1998) Identification of EEG events in the MR scanner: the problem of pulse artifact and a method for its subtraction. Neuroimage 8:229–239. https://doi.org/10.1006/nimg.1998.0361
Allen PJ, Josephs O, Turner R (2000) A method for removing imaging artifact from continuous EEG recorded during functional MRI. Neuroimage 12:230–239. https://doi.org/10.1006/nimg.2000.0599
Bagshaw AP, Hawco C, Bénar CG, Kobayashi E, Aghakhani Y, Dubeau F, Pike GB, Gotman J (2005) Analysis of the EEG-fMRI response to prolonged bursts of interictal epileptiform activity. Neuroimage 24:1099–1112. https://doi.org/10.1016/j.neuroimage.2004.10.010
Beckmann CF, Smith SM (2004) Probabilistic independent component analysis for functional magnetic resonance imaging. IEEE Trans Med Imaging 23:137–152. https://doi.org/10.1109/TMI.2003.822821
Bénar CG, Gross DW, Wang Y, Petre V, Pike B, Dubeau F, Gotman J (2002) The BOLD response to interictal epileptiform discharges. Neuroimage 17:1182–1192. https://doi.org/10.1006/nimg.2002.1164
Bénar CG, Schön D, Grimault S, Nazarian B, Burle B, Roth M, Badier JM, Marquis P, Liegeois-Chauvel C, Anton JL (2007) Single-trial analysis of oddball event-related potentials in simultaneous EEG-fMRI. Hum Brain Mapp 28:602–613. https://doi.org/10.1002/hbm.20289
Bernier M, Cunnane SC, Whittingstall K (2018) The morphology of the human cerebrovascular system. Hum Brain Mapp 39:4962–4975. https://doi.org/10.1002/hbm.24337
Bridwell DA, Wu L, Eichele T, Calhoun VD (2013) The spatiospectral characterization of brain networks: fusing concurrent EEG spectra and fMRI maps. Neuroimage 69:101–111. https://doi.org/10.1016/j.neuroimage.2012.12.024
Bruns A (2004) Fourier-, Hilbert-and wavelet-based signal analysis: are they really different approaches? J Neurosci Methods 137:321–332
Buxton RB, Uludaǧ K, Dubowitz DJ, Liu TT (2004) Modeling the hemodynamic response to brain activation. Neuroimage 23:220–233. https://doi.org/10.1016/j.neuroimage.2004.07.013
Calhoun VD, Adali T, Pearlson GD, Kiehl KA (2006) Neuronal chronometry of target detection: fusion of hemodynamic and event-related potential data. Neuroimage 30(2):544–553
Chang C, Leopold DA, Schölvinck ML, Mandelkow H, Picchioni D, Liu X, Frank QY, Turchi JN, Duyn JH (2016) Tracking brain arousal fluctuations with fMRI. Proc Natl Acad Sci USA 113:4518–4523
Chen T, Ohlsson H, Ljung L (2012) On the estimation of transfer functions, regularizations and Gaussian processes—revisited. Automatica 48:1525–1535
de Munck JC, Gonçalves SI, Huijboom L, Kuijer JPA, Pouwels PJW, Heethaar RM, Lopes da Silva FH, Goncalves SI, Huijboom L, Kuijer JPA, Pouwels PJW, Heethaar RM, Lopes da Silva FH, Gonçalves SI, Huijboom L, Kuijer JPA, Pouwels PJW, Heethaar RM, Lopes da Silva FH (2007) The hemodynamic response of the alpha rhythm: an EEG/fMRI study. Neuroimage 35:1142–1151. https://doi.org/10.1016/j.neuroimage.2007.01.022
de Munck JC, Gonçalves SI, Mammoliti R, Heethaar RM, Lopes da Silva FH (2009) Interactions between different EEG frequency bands and their effect on alpha-fMRI correlations. Neuroimage 47:69–76. https://doi.org/10.1016/j.neuroimage.2009.04.029
Delorme A, Makeig S (2004) EEGLAB: an open source toolbox for analysis of single-trial EEG dynamics including independent component analysis. J Neurosci Methods 134:9–21. https://doi.org/10.1016/j.jneumeth.2003.10.009
Dickie EW, Anticevic A, Smith DE, Coalson TS, Manogaran M, Calarco N, Viviano JD, Glasser MF, Van Essen DC, Voineskos AN (2019) Ciftify: a framework for surface-based analysis of legacy MR acquisitions. Neuroimage 197:818–826. https://doi.org/10.1016/j.neuroimage.2019.04.078
die Jong S (1993) SIMPLS: an alternative approach squares regression to partial least. Chemom Intell Lab Syst 18:251–263. https://doi.org/10.1016/0169-7439(93)85002-X
Ebisch B, Schmidt KE, Niessing M, Singer W, Galuske RAW, Niessing J (2005) Hemodynamic signals correlate tightly with synchronized gamma oscillations. Science 309:948–951
Erbil N, Ungan P (2007) Changes in the alpha and beta amplitudes of the central EEG during the onset, continuation, and offset of long-duration repetitive hand movements. Brain Res 1169:44–56. https://doi.org/10.1016/J.BRAINRES.2007.07.014
Falahpour M, Chang C, Wong CW, Liu TT (2018) Template-based prediction of vigilance fluctuations in resting-state fMRI. Neuroimage 174:317–327
Fischl B (2012) FreeSurfer. Neuroimage. https://doi.org/10.1016/j.neuroimage.2012.01.021
Friston KJ, Fletcher P, Josephs O, Holmes ANDREW, Rugg MD, Turner R (1998) Event-related fMRI: characterizing differential responses. Neuroimage 7(1):30–40
Friston KJ, Mechelli A, Turner R, Price CJ (2000) Nonlinear responses in fMRI: the balloon model, Volterra kernels, and other hemodynamics. Neuroimage 12:466–477. https://doi.org/10.1006/nimg.2000.0630
Fuglø D, Pedersen H, Rostrup E, Hansen AE, Larsson HBW (2012) Correlation between single-trial visual evoked potentials and the blood oxygenation level dependent response in simultaneously recorded electroencephalography-functional magnetic resonance imaging. Magn Reson Med 68:252–260. https://doi.org/10.1002/mrm.23227
Goense JBM, Logothetis NK (2008) Neurophysiology of the BOLD fMRI signal in awake monkeys. Curr Biol 18:631–640. https://doi.org/10.1016/j.cub.2008.03.054
Goldman RI, Stern JM, Engel J, Cohen MS (2002) Simultaneous EEG and fMRI of the alpha rhythm. NeuroReport 13:2487–2492. https://doi.org/10.1097/01.wnr.0000047685.08940.d0
Gómez JC, Baeyens E (2004) Identification of block-oriented nonlinear systems using orthonormal bases. J Process Control 14:685–697. https://doi.org/10.1016/j.jprocont.2003.09.010
Gramfort A, Papadopoulo T, Olivi E, Clerc M (2009) OpenMEEG: opensource software for quasistatic bioelectromagnetics. Biomed Eng Online 8:1. https://doi.org/10.1186/1475-925X-8-1
Heuberger PSC, Van den Hof PMJJ, Wahlberg B (2005) Modeling and identification with rational orthogonal basis functions. Springer, Berlin
Hossein-Zadeh GA, Ardekani BA, Soltanian-Zadeh H (2003) A signal subspace approach for modeling the hemodynamic response function in fMRI. Magn Reson Imaging 21:835–843. https://doi.org/10.1016/S0730-725X(03)00180-2
Jansen M, White TP, Mullinger KJ, Liddle EB, Gowland PA, Francis ST, Bowtell R, Liddle PF (2012) Motion-related artefacts in EEG predict neuronally plausible patterns of activation in fMRI data. Neuroimage 59:261–270. https://doi.org/10.1016/J.NEUROIMAGE.2011.06.094
Jenkinson M, Beckmann CF, Behrens TEJ, Woolrich MW, Smith SM (2012) Fsl. Neuroimage 62:782–790. https://doi.org/10.1016/j.neuroimage.2011.09.015
Jorge J, Van der Zwaag W, Figueiredo P (2014) EEG-fMRI integration for the study of human brain function. Neuroimage 102:24–34. https://doi.org/10.1016/j.neuroimage.2013.05.114
Kilner JM, Mattout J, Henson R, Friston KJ (2005) Hemodynamic correlates of EEG: a heuristic. Neuroimage 28:280–286. https://doi.org/10.1016/j.neuroimage.2005.06.008
Klein A, Tourville J (2012) 101 labeled brain images and a consistent human cortical labeling protocol. Front Neurosci 6:171. https://doi.org/10.3389/fnins.2012.00171
Laufs H, Kleinschmidt A, Beyerle A, Eger E, Salek-Haddadi A, Preibisch C, Krakow K (2003) EEG-correlated fMRI of human alpha activity. Neuroimage 19:1463–1476. https://doi.org/10.1016/S1053-8119(03)00286-6
Laufs H, Holt JL, Elfont R, Krams M, Paul JS, Krakow K, Kleinschmidt A (2006) Where the BOLD signal goes when alpha EEG leaves. Neuroimage 31:1408–1418. https://doi.org/10.1016/j.neuroimage.2006.02.002
Leistedt B, McEwen JD (2012) Exact wavelets on the ball. IEEE Trans Signal Process 60:6257–6269. https://doi.org/10.1109/TSP.2012.2215030
Leite M, Leal A, Figueiredo P (2013) Transfer function between EEG and BOLD signals of epileptic activity. Front Neurol. https://doi.org/10.3389/fneur.2013.00001
Le Van Quyen M, Foucher J, Lachaux JP, Rodriguez E, Lutz A, Martinerie J, Varela FJ (2001) Comparison of Hilbert transform and wavelet methods for the analysis of neuronal synchrony. J Neurosci Methods 111:83–98
Logothetis NK, Pauls J, Augath M, Trinath T, Oeltermann A (2001) Neurophysiological investigation of the basis of the fMRI signal. Nature 412:150–157. https://doi.org/10.1038/35084005
Lu Y, Bagshaw AP, Grova C, Kobayashi E, Dubeau F, Gotman J (2006) Using voxel-specific hemodynamic response function in EEG-fMRI data analysis. Neuroimage 32:238–247
Magri C, Schridde U, Murayama Y, Panzeri S, Logothetis NK (2012) The amplitude and timing of the BOLD signal reflects the relationship between local field potential power at different frequencies. J NeuroSci 32:1395–1407. https://doi.org/10.1523/JNEUROSCI.3985-11.2012
Mantini D, Perrucci MG, Del Gratta C, Romani GL, Corbetta M (2007) Electrophysiological signatures of resting state networks in the human brain. Proc Natl Acad Sci USA 104:13170–13175. https://doi.org/10.1073/pnas.0700668104
Marecek R, Lamos M, Mikl M, Barton M, Fajkus J, Rektor I, Brazdil M (2016) What can be found in scalp EEG spectrum beyond common frequency bands. EEG-fMRI study. J Neural Eng. https://doi.org/10.1088/1741-2560/13/4/046026
Marino M, Liu Q, Koudelka V, Porcaro C, Hlinka J, Wenderoth N, Mantini D (2018) Adaptive optimal basis set for BCG artifact removal in simultaneous EEG-fMRI. Sci Rep 8:1–11. https://doi.org/10.1038/s41598-018-27187-6
Marmarelis VZ (1993) Identification of nonlinear biological systems using Laguerre expansions of kernels. Ann Biomed Eng 21:573–589
Marmarelis VZ (2004) Nonlinear dynamic modeling of physiological systems. Wiley, New York. https://doi.org/10.1002/9780471679370
Moosmann M, Ritter P, Krastel I, Brink A, Thees S, Blankenburg F, Taskin B, Obrig H, Villringer A (2003) Correlates of alpha rhythm in functional magnetic resonance imaging and near infrared spectroscopy. Neuroimage 20:145–158. https://doi.org/10.1016/S1053-8119(03)00344-6
Mullinger KJ, Morgan PS, Bowtell RW (2008) Improved artifact correction for combined electroencephalography/functional MRI by means of synchronization and use of vectorcardiogram recordings. J Magn Reson Imaging 27:607–616. https://doi.org/10.1002/jmri.21277
Mullinger KJ, Yan WX, Bowtell R (2011) Reducing the gradient artefact in simultaneous EEG-fMRI by adjusting the subject’s axial position. Neuroimage 54:1942–1950. https://doi.org/10.1016/j.neuroimage.2010.09.079
Mullinger KJ, Chowdhury MEH, Bowtell R (2014) Investigating the effect of modifying the EEG cap lead configuration on the gradient artifact in simultaneous EEG-fMRI. Front Neurosci 8:1–10. https://doi.org/10.3389/fnins.2014.00226
Murta T, Leite M, Carmichael DW, Figueiredo P, Lemieux L (2015) Electrophysiological correlates of the BOLD signal for EEG-informed fMRI. Hum Brain Mapp 36:391–414. https://doi.org/10.1002/hbm.22623
Murta T, Hu L, Tierney TM, Chaudhary UJ, Walker MC, Carmichael DW, Figueiredo P, Lemieux L (2016) A study of the electro-haemodynamic coupling using simultaneously acquired intracranial EEG and fMRI data in humans. Neuroimage 142:371–380. https://doi.org/10.1016/j.neuroimage.2016.08.001
Muthukumaraswamy SD (2013) High-frequency brain activity and muscle artifacts in MEG/EEG: a review and recommendations. Front Hum Neurosci 7:138. https://doi.org/10.3389/fnhum.2013.00138
Nguyen VT, Cunnington R (2014) The superior temporal sulcus and the N170 during face processing: single trial analysis of concurrent EEG-fMRI. Neuroimage 86:492–502. https://doi.org/10.1016/j.neuroimage.2013.10.047
Ogawa S, Lee TM, Kay AR, Tank DW (1990) Brain magnetic resonance imaging with contrast dependent on blood oxygenation. Proc Natl Acad Sci U S A. https://doi.org/10.1073/pnas.87.24.9868
Ohara S, Mima T, Baba K, Ikeda A, Kunieda T, Matsumoto R, Yamamoto J, Matsuhashi M, Nagamine T, Hirasawa K, Hori T, Mihara T, Hashimoto N, Salenius S, Shibasaki H (2001) Increased synchronization of cortical oscillatory activities between human supplementary motor and primary sensorimotor areas during voluntary movements. J Neurosci 21:9377–9386. https://doi.org/10.1523/JNEUROSCI.21-23-09377.2001
Oldfield RC (1971) The assessment and analysis of handedness. Neuropsychologia 9:97–113
Portnova GV, Tetereva A, Balaev V, Atanov M, Skiteva L, Ushakov V, Ivanitsky A, Martynova O (2018) Correlation of BOLD signal with linear and nonlinear patterns of EEG in resting state EEG-informed fMRI. Front Hum Neurosci 11:1–12. https://doi.org/10.3389/fnhum.2017.00654
Rangaprakash D, Wu G-R, Marinazzo D, Hu X, Deshpande G (2018) Hemodynamic response function (HRF) variability confounds resting-state fMRI functional connectivity. Magn Reson Med 80:1697–1713. https://doi.org/10.1002/mrm.27146
Ritter P, Moosmann M, Villringer A (2009) Rolandic alpha and beta EEG rhythms’ strengths are inversely related to fMRI-BOLD signal in primary somatosensory and motor cortex. Hum Brain Mapp 30:1168–1187. https://doi.org/10.1002/hbm.20585
Rosa MJ, Daunizeau J, Friston KJ (2010a) EEG-fMRI integration: a critical review of biophysical modeling and data analysis approaches. J Integr Neurosci 9:453–476. https://doi.org/10.1142/S0219635210002512
Rosa MJ, Kilner J, Blankenburg F, Josephs O, Penny W (2010b) Estimating the transfer function from neuronal activity to BOLD using simultaneous EEG-fMRI. Neuroimage 49:1496–1509. https://doi.org/10.1016/j.neuroimage.2009.09.011
Rosa MJ, Kilner JM, Penny WD (2011) Bayesian comparison of neurovascular coupling models using EEG-fMRI. PLoS Comput Biol 7:e1002070. https://doi.org/10.1371/journal.pcbi.1002070
Rospiral R, Kramer N (2006) Overview and recent advances in partial least squares 43–51. doi: https://doi.org/10.1075/aals.6.03ch3
Ryali S, Glover GH, Chang C, Menon V (2009) Development, validation, and comparison of ICA-based gradient artifact reduction algorithms for simultaneous EEG-spiral in/out and echo-planar fMRI recordings. Neuroimage 48:348–361. https://doi.org/10.1016/j.neuroimage.2009.06.072
Sato JR, Rondinoni C, Sturzbecher M, de Araujo DB, Amaro E Jr (2010) From EEG to BOLD: brain mapping and estimating transfer functions in simultaneous EEG-fMRI acquisitions. Neuroimage 5:1416–1426
Scheeringa R, Bastiaansen MCM, Petersson KM, Oostenveld R, Norris DG, Hagoort P (2008) Frontal theta EEG activity correlates negatively with the default mode network in resting state. Int J Psychophysiol 67:242–251. https://doi.org/10.1016/j.ijpsycho.2007.05.017
Scheeringa R, Fries P, Petersson KM, Oostenveld R, Grothe I, Norris DG, Hagoort P, Bastiaansen MCM (2011) Neuronal dynamics underlying high- and low-frequency EEG oscillations contribute independently to the human BOLD signal. Neuron 69:572–583. https://doi.org/10.1016/j.neuron.2010.11.044
Scheeringa R, Koopmans PJ, van Mourik T, Jensen O, Norris DG (2016) The relationship between oscillatory EEG activity and the laminar-specific BOLD signal. Proc Natl Acad Sci USA 113:6761–6766. https://doi.org/10.1073/pnas.1522577113
Sclocco R, Tana MG, Visani E, Gilioli I, Panzica F, Franceschetti S, Cerutti S, Bianchi AM (2014) EEG-informed fMRI analysis during a hand grip task: estimating the relationship between EEG rhythms and the BOLD signal. Front Hum Neurosci 8:186. https://doi.org/10.3389/fnhum.2014.00186
Shmuel A, Leopold DA (2008) Neuronal correlates of spontaneous fluctuations in fMRI signals in monkey visual cortex: implications for functional connectivity at rest. Hum Brain Mapp 29:751–761. https://doi.org/10.1002/hbm.20580
Tadel F, Baillet S, Mosher JC, Pantazis D, Leahy RM (2011) Brainstorm: a user-friendly application for MEG/EEG analysis. Comput Intell Neurosci. https://doi.org/10.1155/2011/879716
Thornton RC, Rodionov R, Laufs H, Vulliemoz S, Vaudano A, Carmichael D, Cannadathu S, Guye M, McEvoy A, Lhatoo S, Bartolomei F, Chauvel P, Diehl B, De Martino F, Elwes RDC, Walker MC, Duncan JS, Lemieux L (2010) Imaging haemodynamic changes related to seizures: comparison of EEG-based general linear model, independent component analysis of fMRI and intracranial EEG. Neuroimage 53:196–205. https://doi.org/10.1016/j.neuroimage.2010.05.064
Tyvaert L, LeVan P, Grova C, Dubeau F, Gotman J (2008) Effects of fluctuating physiological rhythms during prolonged EEG-fMRI studies. Clin Neurophysiol 119:2762–2774. https://doi.org/10.1016/j.clinph.2008.07.284
Uji M, Wilson R, Francis ST, Mullinger KJ, Mayhew SD (2018) Exploring the advantages of multiband fMRI with simultaneous EEG to investigate coupling between gamma frequency neural activity and the BOLD response in humans. Hum Brain Mapp 39:1673–1687
Van Veen BD, Van Drongelen W, Yuchtman M, Suzuki A (1997) Localization of brain electrical activity via linearly constrained minimum variance spatial filtering. IEEE Trans Biomed Eng 44:867–880. https://doi.org/10.1109/10.623056
van Wijk BCM, Beek PJ, Daffertshofer A (2012) Differential modulations of ipsilateral and contralateral beta (de)synchronization during unimanual force production. Eur J Neurosci 36:2088–2097. https://doi.org/10.1111/j.1460-9568.2012.08122.x
Wager TD, Nichols TE (2003) Optimization of experimental design in fMRI: a general framework using a genetic algorithm. Neuroimage 182:293–309. https://doi.org/10.1016/S1053-8119(02)00046-0
Wan X, Riera J, Iwata K, Takahashi M, Wakabayashi T, Kawashima R (2006) The neural basis of the hemodynamic response nonlinearity in human primary visual cortex: implications for neurovascular coupling mechanism. Neuroimage 32:616–625. https://doi.org/10.1016/j.neuroimage.2006.03.040
Westwick DT, Kearney RE (2003) Identification of nonlinear physiological systems. Wiley, New York
Wirsich J, Bénar C, Ranjeva JP, Descoins M, Soulier E, Le Troter A, Confort-Gouny S, Liégeois-Chauvel C, Guye M (2014) Single-trial EEG-informed fMRI reveals spatial dependency of BOLD signal on early and late IC-ERP amplitudes during face recognition. Neuroimage 100:325–336. https://doi.org/10.1016/j.neuroimage.2014.05.075
Wu G-R, Liao W, Stramaglia S, Ding J-R, Chen H, Marinazzo D (2013) A blind deconvolution approach to recover effective connectivity brain networks from resting state fMRI data. Med Image Anal 17:365–374. https://doi.org/10.1016/j.media.2013.01.003
Xifra-Porxas A, Niso G, Larivière S, Kassinopoulos M, Baillet S, Mitsis GD, Boudrias M-H (2019) Older adults exhibit a more pronounced modulation of beta oscillations when performing sustained and dynamic handgrips. Neuroimage 201:116037. https://doi.org/10.1016/J.NEUROIMAGE.2019.116037
Funding
This work was supported by funds from the Natural Sciences and Engineering Research Council of Canada (NSERC) Discovery Grants RGPIN-2019-06638 [GDM], and RGPIN-2017-05270 [MHB], the Fonds de la Recherche du Quebec—Nature et Technologies (FRQNT) Team Grant 254680-2018 [GDM], the Canadian Foundation for Innovation grant numbers 34362 [GDM] and 34277 [MHB]. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
PCP: Conceptualization, Investigation, Data curation, Methodology, Software, Formal analysis, Visualization, Writing—original draft. AX-P: Investigation, Data curation, Methodology, Formal analysis, Writing—reviewing and editing. MK: Investigation, Data curation, Formal analysis, Writing—reviewing and editing. M-HB: Conceptualization, Methodology, Resources, Writing—reviewing and editing, Funding acquisition. GDM: Conceptualization, Methodology, Resources, Writing—reviewing and editing, Project administration, Funding acquisition, Supervision.
Corresponding author
Ethics declarations
Conflict of interest
Not applicable.
Ethical Approval
This study was performed in accordance with the McGill University Ethical Advisory Committee.
Consent to Participate
Informed consent was obtained from all individual participants included in the study.
Additional information
Handling Editor: Louis Lemieux.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Appendices
Appendix
Identification of Hammerstein and Wiener-Hammerstein Models
The Hammerstein model (Fig. 1a; Eq. (1)) can be estimated efficiently from the input–output data using orthonormal basis functions for the representation of the LTI block (Gómez and Baeyens 2004), which is given by
where \(\left\{{\text{B}}_{\text{j}}\left(\text{n}\right);\text{j}=0,\dots ,\text{ L}-1;\text{n}=0,\dots ,\text{M}\right\}\) is a set of \(\text{L}\) orthonormal basis functions, and \({\text{b}}_{\text{j}}\) is the unknown expansion coefficient of the \(\text{j}\)-th order basis function. The use of orthonormal bases reduces the number of required free parameters in the model and allows parameter estimation using least-squares regression. This leads to increased estimation accuracy in the presence of noise even from short experimental data-records. Combining Eqs. (1, 2) and (6), the input–output relationship can be written as
where \({\text{v}}_{\text{i},\text{j}}^{\left(\text{p}\right)}\left(\text{n}\right)\) denotes the convolution between the \(\text{p}\)-th polynomial power of the \(\text{i}\)-th input with the \(\text{j}\)-th basis function. Equation (7) can be re-expressed as a linear regression problem
where \(\mathbf{V}=\left[{\text{v}}_{\text{1,0}}^{\left(1\right)},\dots , {\text{v}}_{\text{Q},0}^{\left(1\right)}, \dots ,{\text{v}}_{1,\text{L}-1}^{\left(1\right)},\dots ,{\text{v}}_{\text{Q},\text{L}-1}^{\left(1\right)} ,\dots \dots , {\text{v}}_{\text{1,0}}^{\left(\text{P}\right)},\dots , {\text{v}}_{\text{Q},0}^{\left(\text{P}\right)}, \dots ,{\text{v}}_{1,\text{L}-1}^{\left(\text{P}\right)},\dots ,{\text{v}}_{\text{Q},\text{L}-1}^{\left(\text{P}\right)}\right]\), and \({\mathbf{c}} = \left[ {{\text{a}}_{{1,1}} {\text{b}}_{0} , \ldots ,{\text{a}}_{{1,1}} {\text{b}}_{{{\text{L}} - 1}} , \ldots ,{\text{a}}_{{{\text{Q}},1}} {\text{b}}_{0} , \ldots ,{\text{a}}_{{{\text{Q}},1}} {\text{b}}_{{{\text{L}} - 1}} , \ldots \ldots ,{\text{a}}_{{1,{\text{P}}}} {\text{b}}_{0} , \ldots ,{\text{a}}_{{1,{\text{P}}}} {\text{b}}_{{{\text{L}} - 1}} , \ldots ,{\text{a}}_{{{\text{Q,P}}}} {\text{b}}_{0} , \ldots ,{\text{a}}_{{{\text{Q,P}}}} {\text{b}}_{{{\text{L}} - 1}} } \right]^{{\text{T}}}\) is a vector of the unknown model parameters.
Power fluctuations within distinct EEG frequency bands are highly correlated as previously reported in the literature (de Munck et al. 2009). As a result, the columns in \(\mathbf{V}\) are strongly collinear, which makes estimation of \(\mathbf{c}\) using ordinary least-squares numerically unstable due to ill-conditioning of the Gram matrix \(\left[{\mathbf{V}}^{\mathbf{T}}\mathbf{V}\right]\). Therefore, to obtain a numerically more stable estimate of the unknown parameter vector \(\mathbf{c}\), we employed partial least-squares regression (PLSR) (Rospiral and Kramer 2006).
PLSR is performed in three phases. In phase 1, the algorithm finds projections of \(\mathbf{V}\) and \(\mathbf{y}\) to a new co-ordinate system such that the covariance of these projections is maximized. This is achieved using a linear decomposition of both \(\mathbf{V}\) and \(\mathbf{y}\) into a set of orthonormal latent variables (scores) and loadings given by
where \(\mathbf{T}\) and \(\mathbf{U}\) are matrices of latent variables associated with \(\mathbf{V}\) and \(\mathbf{y}\), respectively. \(\mathbf{P}\) and \(\mathbf{W}\) are the corresponding loadings for each latent variable matrix, and \({\mathbf{e}}_{1,2}\) are error terms. The decomposition of \(\mathbf{V}\) and \(\mathbf{y}\) is performed such that the covariance between \(\mathbf{T}\) and \(\mathbf{U}\) is maximized. In phase 2, the algorithm performs ordinary least-squares regression analysis between the latent variables \(\mathbf{T}\) and system output \(\mathbf{y}\)
where \({\varvec{\uptheta}}={\left[{\uptheta }_{1},\dots , {\uptheta }_{\text{Q}\times \text{L}\times \text{P}}\right]}^{\text{T}}\) is a vector of the regression coefficients. Note that in this case the Gram matrix \(\left[{\mathbf{T}}^{\mathbf{T}}\mathbf{T}\right]\) is well-conditioned since the columns in \(\mathbf{T}\) are orthonormal. In phase 3, the estimated \({\widehat{{\varvec{\uptheta}}}}_{\mathbf{L}\mathbf{S}\mathbf{E}}\) coefficients are projected back to the original parameter space yielding unbiased estimates of the original model parameters \({\widehat{\mathbf{c}}}_{\mathbf{P}\mathbf{L}\mathbf{S}}\) (die Jong 1993).
To uniquely identify the unknown parameters of the Hammerstein model described by (1, 2) and (6, 7), the bilinear parameter vector \(\mathbf{c}\) needs to be dissociated into its constituent \({\text{a}}_{\text{i},\text{p}}\) and \({\text{b}}_{\text{j}}\) parameters. The parameter vector \(\mathbf{c}\) can be reshaped into a block-column matrix \({\mathbf{c}}_{\mathbf{a}\mathbf{b}}\), such that
where \(\mathbf{a}={\left[{\text{a}}_{\text{1,1}},\dots ,{\text{a}}_{\text{Q},1},{\text{a}}_{\text{1,2}},\dots \dots , {\text{a}}_{\text{Q},\text{P}-1}, {\text{a}}_{1,\text{P}},\dots ,{\text{a}}_{\text{Q},\text{P}}\right]}^{\text{T}}\) and \(\mathbf{b}={\left[{\text{b}}_{0},\dots ,{\text{b}}_{\text{L}-1}\right]}^{\text{T}}\). An optimal, in the least-squares sense, estimate of the model parameters \({\widehat{\mathbf{a}}}_{\mathbf{L}\mathbf{S}\mathbf{E}}\) and \({\widehat{\mathbf{b}}}_{\mathbf{L}\mathbf{S}\mathbf{E}}\) can be obtained solving the following constrained minimization problem
where \(\text{h}\left(\text{m}\right)\) is given by (6). Note that as a result of normalizing the polynomial coefficients \(\mathbf{a}\), the estimate \({\widehat{\mathbf{a}}}_{\mathbf{L}\mathbf{S}\mathbf{E}}\) reflects a relative rather than absolute contribution of individual EEG bands to the BOLD signal variance. A solution to (A9) is provided by the singular value decomposition (SVD) of matrix \({\mathbf{c}}_{\mathbf{a}\mathbf{b}}\) (Gómez and Baeyens 2004). Specifically,
where \({\mathbf{U}}_{1}\) is the first left singular vector, \({\mathbf{V}}_{1}\) the first right singular vector, and \({{\varvec{\Sigma}}}_{1}\in {\mathbb{R}}\) the first singular value of the SVD of \({\widehat{\mathbf{c}}}_{\text{ab}}\).
Hammerstein-Wiener Model Identification
The MISO Hammerstein-Wiener (HW) described by Eq. (3) can be re-expressed in a compact matrix form as
where \(\mathbf{F}\) denotes a matrix the columns of which are polynomial powers of \({\text{y}}_{\text{H}}\), and \(\mathbf{z}\) is a vector of the unknown polynomial coefficients, which can be estimated using ordinary least-squares
Orthonormal Basis Functions
There are several sets of orthonormal basis functions that can be used for modeling the impulse response function of the LTI block in the Hammerstein and Wiener-Hammerstein model configuration (Heuberger et al. 2005). The selection of the appropriate basis set depends on the dynamic behavior of the system to be modelled. One basis set that has been extensively used in the literature for modeling of physiological systems is the Laguerre basis. Laguerre basis functions exhibit exponentially decaying structure and constitute an orthonormal set in \(\left[0,\infty \right)\), which makes them suitable for modeling causal systems with finite memory (Marmarelis 1993).
In this work we employ a smoother variant of the Laguerre basis functions that is based on the spherical Laguerre basis functions (Leistedt and McEwen 2012), which allow obtaining robust HRF estimates in single voxels even during resting conditions where the signal-to-noise ratio (SNR) is particularly low. The \(\text{j}\)-th spherical Laguerre basis function \({\text{b}}_{\text{j}}\left(\text{n}\right); \text{j}=0,\dots ,\text{L}-1;\text{ n}=1,\dots ,\text{M}\) is given by
where \(\alpha\in {\mathbb{R}}_{+}\) is a parameter that determines the rate of exponential asymptotic decline of \({\text{b}}_{\text{j}}\left(\text{n}\right)\), and \({\text{K}}_{\text{j}}\left(\text{n}\right)\) is the \(\text{j}\)-th generalized Laguerre polynomial of order two, defined as
The \(\text{j}\)-th spherical Laguerre basis function \({\text{b}}_{\text{j}}\left(\text{n}\right)\) were convolved with a Gaussian kernel G(μ,τ), with μ > 0 and τ = 1. The μ > 0 parameter controls how late \({\text{b}}_{\text{j}}\left(\text{n}\right)\) will start to fluctuate. The range of this parameter was set to 2.5 < μ < 5 corresponding to HRF estimates with time-to-peak ranging between 3 and 10 s (Fig. S12). The orthonormal properties of the derived basis set was ensured by entering the result of this convolution into an orthogonalization process based on the Gram-Schmidt orthogonalization algorithm.
Rights and permissions
About this article
Cite this article
Prokopiou, P.C., Xifra-Porxas, A., Kassinopoulos, M. et al. Modeling the Hemodynamic Response Function Using EEG-fMRI Data During Eyes-Open Resting-State Conditions and Motor Task Execution. Brain Topogr 35, 302–321 (2022). https://doi.org/10.1007/s10548-022-00898-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10548-022-00898-w