It's a well-known fact that soda is bad for your health. We've all read the reports and articles detailing how the sugar in pop will rot our teeth, cause obesity, or give us diabetes. So perhaps, like many other people, you've turned to carbonated water to fill that cold, bubbly niche in your life. Companies like La Croix or Perrier have grown immensely in recent years, signalling a rising popularity in soda waters- sometimes flavoured, often calorie-free, ‘healthy’ alternative to Coke or Pepsi.
But new reports with headlines like ‘Sparkling Water Is ‘Extremely Acidic’ And Can Ruin Teeth, Dentist Warns’, ‘Tragic News for People Who Love Seltzer Water’, and my personal favourite ‘Flavored waters — yes, including La Croix — are eroding your teeth’ are casting doubt on bubbly water’s healthy status. With claims like ‘seltzers can be bad for your teeth, especially if they are flavoured’ it’s becoming less clear what's good for our teeth.
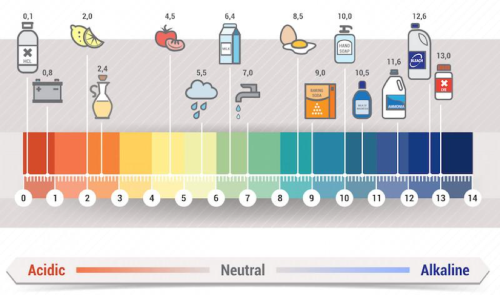
I decided to put some carbonated water to the test, specifically the potential of hydrogen test. The pH of a substance tells us its acidity or basicity- water is roughly neutral with a pH of 7. Here’s a handy reference sheet to give you some context.
I spent a day in the lab and took a total of 36 pH measurements on 9 different brands of carbonated water that I looked very strange buying at IGA. I opted to test each bottle both cold (4℃, the usual temperature for a fridge) and warm (21℃), and both carbonated and decarbonated. You can see my full results in the chart below.
|
pH tests |
|
|
|
|
|
Water Brand |
Warm, carbonated pH |
Cold, carbonated pH |
Warm, decarbonated pH |
Cold, decarbonated pH |
|
Sensations (Lemon) |
5.52 |
4.6 |
6.33 |
5.67 |
|
Saint-Justin |
5.75 |
4.66 |
5.95 |
5.56 |
|
Badoit |
5.88 |
5.12 |
6.27 |
5.46 |
|
Gerolsteiner |
5.82 |
5.2 |
6.35 |
5.89 |
|
Nestle (Pure Life) |
5.13 |
4.45 |
5.8 |
5.5 |
|
Club Soda |
4.4 |
3.69 |
4.99 |
4.77 |
|
Perrier |
5.25 |
4.68 |
6.03 |
5.11 |
|
San Pellegrino |
5.34 |
4.28 |
6.85 |
5.6 |
|
Eska (Lemon) |
4.95 |
4.05 |
5.43 |
5.63 |
The really important numbers to look at are the ones for cold, carbonated waters, as this is how most people will drink their bubbly water. The average for all cold, carbonated water’s pH is 4.5. There seems to be an idea circulating that unflavoured bubbly waters are actually ok for your teeth, but flavoured ones do damage. That idea isn’t exactly reflected in my data, though I did only test 2 flavoured waters (and both lemon since that’s my favourite). The Eska brand lemon water came out as more acidic, but still rather close to the average, and the Sensations brand actually tested less acidic than some of the unflavoured waters. I’d have to do more tests to be sure, but it seems that there isn’t much truth to the idea that it’s the flavours causing tooth damage.
According to several sources, teeth begin to demineralize at a pH of 5.5, but if you look closely at this issue it’s not so clear cut. The Journal of the Canadian Dental Association (JCDA) published this issue in 2003, which looks at the idea of a ‘critical pH’, or the pH below which your teeth are in danger of erosion. As they put it, ‘the critical pH does not have a fixed value but rather is inversely proportional to the calcium and phosphate concentrations in the solution’.
Essentially, dental enamel is made mostly of hydroxyapatite, which dissolves in water to form calcium, phosphate and hydroxyl (OH-) ions. So when we drink liquids without calcium or phosphate ions in them, or with decreased hydroxyl ion amounts (such as acidic solutions) some amount of the minerals from our teeth dissolve into the liquid (the universe likes to balance things as much as it can). This occurs every single time you drink water, but only in tiny amounts (about 30 mg in 1 L of water), since the pH of water is neutral. When we drink acidic drinks, like sodas, fruit juices, or (mildly) acidic bubbly waters, the minerals in our teeth dissolve in a process called demineralization.
So how acidic is too acidic then? When is this equilibrium between hydroxyapatite and dissolved minerals tipped too far? Well, that depends on the pH of your saliva. If you have a pool, you may just have litmus strips sitting around, and it could be fun to find the pH of your spit. But if you don’t, take my word for it that saliva pH is ~5.5-6.5, depending on the phosphate and calcium ions in your body. The more ions in your spit, the lower the critical pH, or in other words, the more acid your teeth can withstand before demineralizing.
So, long story short, are carbonated waters good for your teeth? No. But they’re not as bad as most of the other things we drink. The pH of most bubbly waters might be below the critical pH for most mouths, but as a chemistry student I’d say the claims that these waters are ‘extremely acidic’ are completely false. The pH of Gatorade is about 3, of orange juice is about 3-4 and of Coke is 2.4 (Read about the pH’s of other common drinks here). If the pH to stay above is ~6, then drinking your daily Perrier warm, or even better warm and flat would be best. But if like me, you think that defeats the purpose, then do what I do and rinse your mouth after with tap water.
It might not be that seltzer water is quite the healthy alternative to soda we all hoped, but a better alternative it certainly is. Skip the Pepsi and quench the craving with some S. Pellegrino, your teeth will thank you, and your blood sugar will too. If this foray in the science of seltzer has taught me anything though, it’s that I need to drink more plain old, flat water.







